 Crookes tubes were the first cold cathode vacuum tubes, and they worked by ionising the remaining gas atoms in the tube between the cathode and the anode using a high electrical potential of thousands of volts. To bring the electrons into the tube, they must first be isolated from the atoms in the cathode. In 1927, Germer and Davisson used a crystal lattice to specifically illustrate the cathode rays' wave-like behaviour.Ĭathode rays are so-named due to the reason they are emitted by the negative cathode or electrode in a vacuum tube. Later in 1924, Louis de Broglie showed in his doctoral dissertation that, in fact, electrons are much like photons in the respect that they act both as particles and as waves in a dual manner as Albert Einstein had shown for light earlier. Also, these can create fluorescence, mechanical effects, and more. Since scientists knew it was difficult to deflect electromagnetic waves with an electric field, this was evidence that the beams were made up of particles. This debate between them was resolved when an electric field was used to deflect the rays by J. Also, Crookes insisted it was a particle, while Hertz maintained it as a wave. These conflicting properties have caused disruptions when trying to classify it either as a particle or wave. Ernest Rutherford has demonstrated that the rays could pass through the thin metal foils, the behaviour expected of a particle. Like a wave, the cathode rays travel in straight lines and form a shadow when obstructed by the objects. This particular effect, when coupled with the controlled deflection of a cathode ray either by a magnetic or electric field, the cathode-ray oscilloscope (rather than the cathode-ray tube CRT) is used to track the values and changes of an alternating current or voltage, as well as the image tube used in radar and television. When cathode rays collide with particular molecules used to cover a cathode plate, the molecules (and thus the screen) emit light. X-rays or a very high temperature are created by cathode rays centred on a hard target (anticathode) on a small object in a vacuum (cathode-ray furnace). 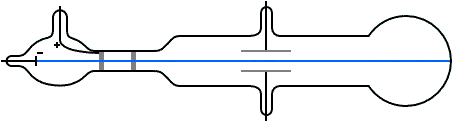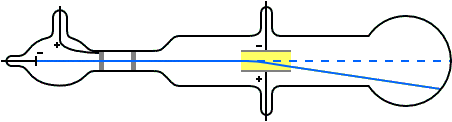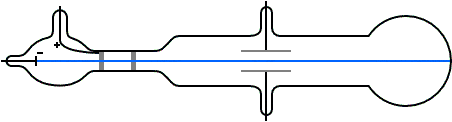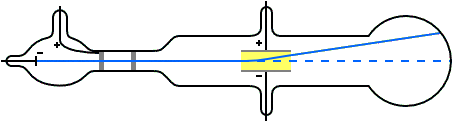
It explained the neutrality of atom, but can not explain the stability of atom.A cathode ray is defined as a stream of electrons leaving the negative electrode (or cathode) in a discharge tube that contains gas at low pressure or the electrons emitted by the heated filament in certain electron tubes. Thomson atomic modelĪtom considered as a positive charged sphere, electron are distributed uniformly in this sphere. It cannot explain the line spectrum of hydrogen. Rutherford failed to explain Maxwell electrons Releases energy then finally it will merge in nucleus, so Rutherford felled toĮxplain existence of atom. Particle rotates around another charged particle in accelerated motion, then if Electron is continuously changing itsĭirection, that means electron is accelerated motion. Nucleus is different concentric circus, culled orbits. Is concentrated in a small volume called nucleus. Few α-particle are deviatedĬharge in equal amount atom is neutral. Most of the α-particles passed un-deviated through the goldįoil. He mode a circular fluorescent-screenĪround gold foil. 
Heīombarded α-particle on thin gold foil. Gold foil for this experiment, because gold foil is most malleable. Mass of neutron = 1.675 □ 10¯^27 kg and charge of neutron = Zero Rutherford Atomic Model (α-particle scattering experiment) Mass of Elementary particles (Important Data) It is a neutral particle, it is produced by bombardment of α-particle on Deflected in electric field: Anode ray are anode rays travel in straight line like cathode rays. Nature of anode rays (charge / mass ratio)ĭepends. In the charge to mass ratio is different. therefore, anode rays are not generated from anode. Originated from anode when voltage is applied e¯ are emitted from cathode andįilled and +Ve charge ions are formed. cathode rays originated from cathode but anode rays are not Small holes in cathode red glow behind cathode it seems rays are coming fromĪnode to cathode. (II) Discovery of Proton (anode ray experiments) 
Nature of cathode rays do not depend on gas filled in glass tube. 
Nature of cathode ray means charge / mass ratio of cathode rays. Nature of cathode rays.īattery to cathode and these electrons move from cathode to anode in from ofĬathode rays. It confirms that cathode rays move in straight line. create a sharp shadow of object kept in direction of cathode Direction of cathode rays: wheel rotates. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
